  
Similarly to the Hammett ρ value, the magnitude of δ will reflect to what extent a reaction is influenced by steric effects: Log ( k s k CH 3 ) = ρ ∗ σ ∗ + δ E s Ī plot of the ratio of the rates versus the E s value for the substituent will give a straight line with a slope equal to δ. While the Hammett equation accounts for how field, inductive, and resonance effects influence reaction rates, the Taft equation also describes the steric effects of a substituent. Taft in 1952 as a modification to the Hammett equation. The Taft equation is a linear free energy relationship (LFER) used in physical organic chemistry in the study of reaction mechanisms and in the development of quantitative structure–activity relationships for organic compounds. 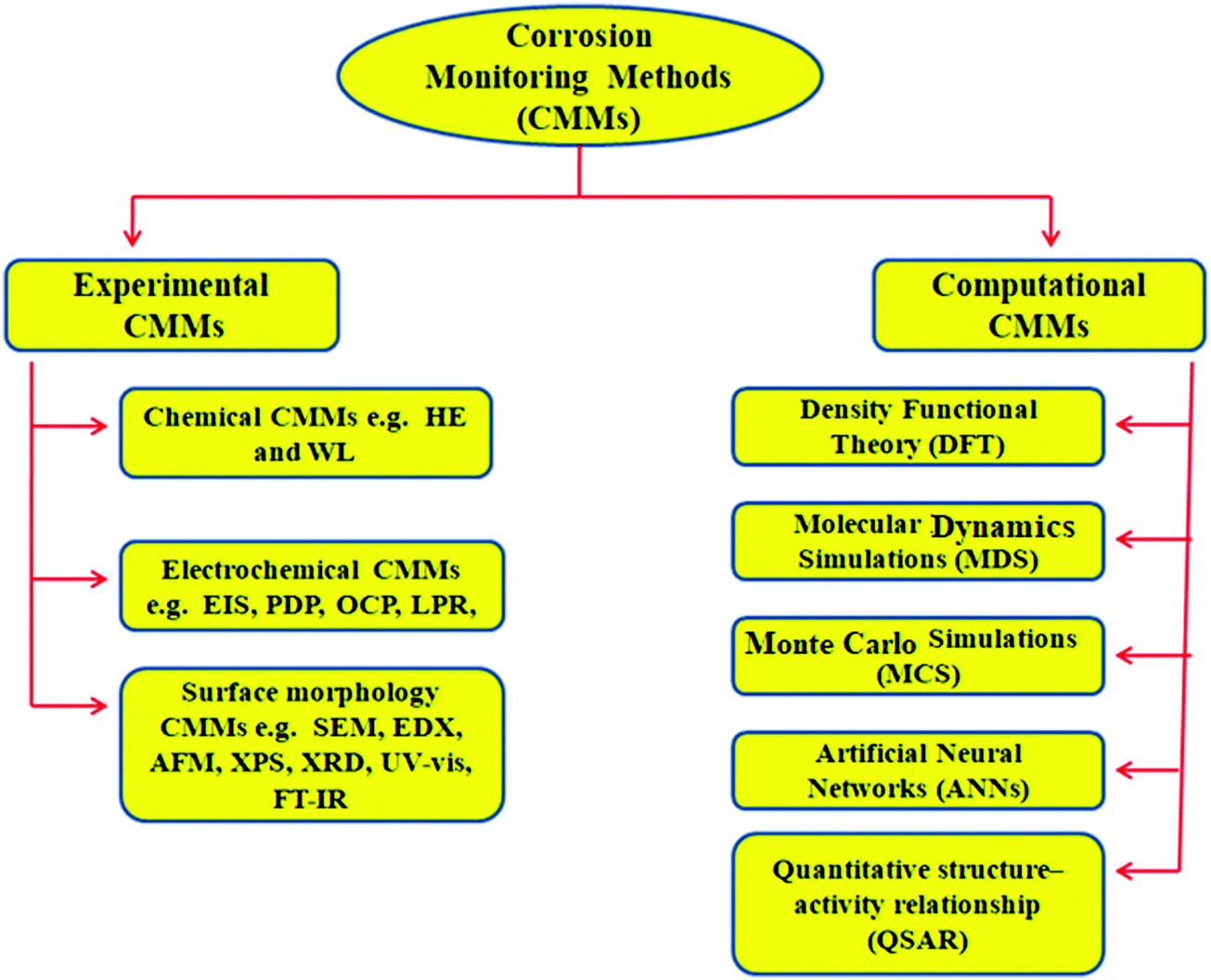
Constants used in the Taft equation Substituent 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
